Scientific Calendar October 2020
How does targeted axillary dissection (TAD) improve staging after neoadjuvant chemotherapy?
When performing TAD, the lymph nodes most likely to harbour metastasis can be identified by visual means only.
TAD means that most of the lymph nodes are removed in order to improve staging.
TAD combines a tracer-guided SLNB with removal of the previously marked positive lymph node and thus reduces the false-negative rate after neoadjuvant chemotherapy.
Congratulations!
That's the correct answer!
Sorry! That´s not completely correct!
Please try again
Sorry! That's not the correct answer!
Please try again
Notice
Please select at least one answer
Scientific background
Neoadjuvant systemic therapy (NAST) has become more popular for treating breast cancer patients. In contrast to the adjuvant systemic therapy, NAST is applied before patients undergo any intervention of cancer surgery. The aim is to downstage or eradicate the disease and thus enable breast-conserving surgery instead of mastectomy. In case of a clinically node-positive (cN+) diagnosis, administration of NAST targets to eradicate the cancer in the lymph nodes, thereby potentially enabling omission of axillary lymph node dissection (ALND) and reducing the risk of post-surgical morbidities such as lymphoedema. Pathologic complete response (pCR) is currently considered the most significant indicator of the success of NAST and is evaluated in the tumour as well as in the axillary lymph nodes.
To enable accurate assessment of the response in the breast and axilla, it is essential that both the positive lymph node(s) and the breast lesion are marked before NAST to be able to locate them correctly afterwards.
How can axillary staging be improved in breast cancer after NAST?
In case the patient shows a positive response to NAST, a less radical surgical approach – such as SLNB instead of ALND – is feasible.
However, the effectiveness of SLNB after NAST has been discussed controversially since systemic therapy impacts the lymphatic system, which might lead to hampered tracer migration. The main concern are false-negative rates (FNR) of over 10% and a low lymph node yield with the risk that cancerous lymph nodes are missed, leading to wrong staging and subsequent wrong treatment (prospective multicentre trials: ACOSOG Z1071, SENTINA, SN FNAC).
A first countermeasure to decrease the FNR was the use of a dual tracer approach (e.g. radioisotope + blue dye) and the excision of more than 2 SLNs. Many international studies showed how the use of a second tracer in association with the standard technique (Tc99) improved the number of captured nodes (> 3) and consequently lowered the FNR. In the meantime, first results have been produced demonstrating the non-inferiority of Magtrace® magnetic tracer as alternative to this standard technique. [1]
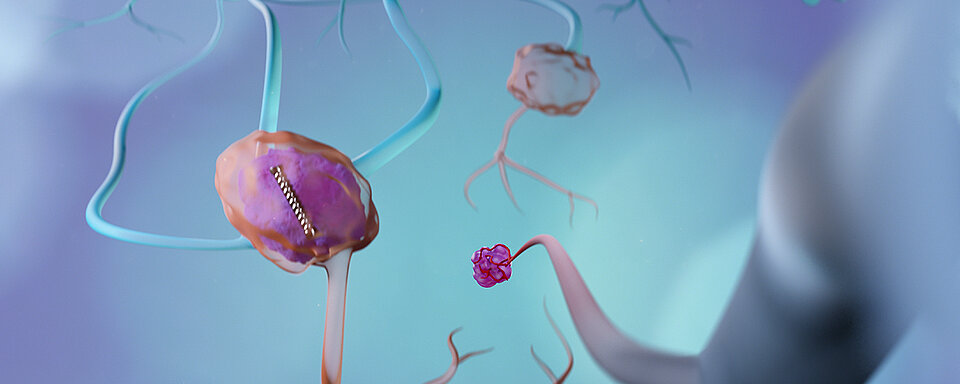
A new promising trend to assess the axilla more accurately includes marking the biopsy-proven metastatic lymph node before NAST in order to securely locate it afterwards. When SLNB is paired with a removal of the previously positive target lymph node, a procedure called targeted axillary dissection (TAD), the operation was shown to become a lot more accurate. Studies of TAD are reporting an FNR of < 2% compared to SLNB alone. [2] This technique is currently investigated further to generate more evidence.
Once detected and excised, all lymph nodes are examined for the presence of metastases. ALND can only be avoided if no residual cancer burden can be detected in the lymph nodes. Therefore, accurate staging is essential and requires a highly sensitive and comprehensive analysis such as is possible with OSNA (one-step nucleic acid amplification). OSNA is a rapid molecular assay which, in contrast to histopathology, allows an analysis of the entire lymph node, thereby avoiding missing even tiny metastases such as are expected particularly after NAST. Furthermore, OSNA results are available already intraoperatively which allows surgeons a fully informed decision during the surgical procedure. This helps sparing patients potential second surgeries in case postoperative histopathology would reveal residual nodal disease.
Why is it difficult to locate previously positive lymph nodes after NAST with confidence?
Through NAST, nodal shrinkage may occur and in consequence, the marker device may shift into surrounding tissue. [3] Further to that, a lack of accuracy in detection is caused by poor or no visibility of the marker due to fibrosis induced by the systemic treatment. When ultrasound is performed intraoperatively, the quality of the localisation results is highly operator-dependent. Thus, difficulties to reliably locate the marker during surgery might occur.
A marker is needed that stays securely in place and can be identified during surgery with an easy detection method, ensuring safe and accurate localisation of the previously positive lymph node after NAST.
One solution for that is the Magseed® magnetic marker, which is CE-marked and FDA-cleared for long-term placement in soft tissue. Clinical studies involving over 200 patients globally have demonstrated accurate placement, no migration once implanted and accurate removal. In addition to improving clinical outcomes, it also offers a better patient experience and promotes seamless radiology and OR scheduling.
When the Magseed® marker is used in combination with the lymphatic tracer Magtrace®, they offer the world’s only wire-free, radiation-free solution for TAD.
References
[1] IMAGINE II (to be published 2020)
[2] Caudle et al. (2016): J Clin Oncol. 34: 1072–8.
[3] Hartmann et al. (2018): EJSO. 44:1307–11.
Endomag®, Sentimag® and Magseed® are registered European Union trade marks of Endomagnetics Ltd. www.endomag.com
Magtrace® is a registered trade mark of Endomagnetics Ltd in the United Kingdom · www.endomag.com